 Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). Hands-on Periodic board Labeled periodic tables. Atomic mass of select elements (along with which rounding off values) is mentioned in the plot below. However when talking about a mole of an ionic compound we will still use the term molar mass. Atomic mass of every elements (along with the rounded off values) is mentioned in that chart below. This is because there are no individual molecules in ionic compounds. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. The molar mass of the N 2 molecule is therefore 28.02 g/mol. In the periodic table, the vertical columns are called groups and the horizontal rows are called periods. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. Periodic Table of Elements - The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. 
For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Substance (formula) Atomic, Molecular, or Formula Mass (amu) Molar Mass (g/mol) carbon (C) 12.011 (atomic mass) 12.011: ethanol (C 2 H 5 OH) 46. The concept of molar mass can also be applied to compounds. 15 Nitrogen 7 16 Oxygen 8 17 Fluorine 9 4.00 - Neon not round them further when performing calculations. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. When the chemical elements are thus arranged, there is a recurring pattern called the periodic law in their properties, in which elements in the same column (group. Thus, the molar mass of bilirubin can be expressed as 584.05 g/mol, which is read as five hundred eighty four. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. The division sign (/) implies per, and 1 is implied in the denominator. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. Because molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). Use this visual tool for calculating molar mass for any chemical formula. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. We have defined a mole based on the isotopic atomic mass of carbon-12. 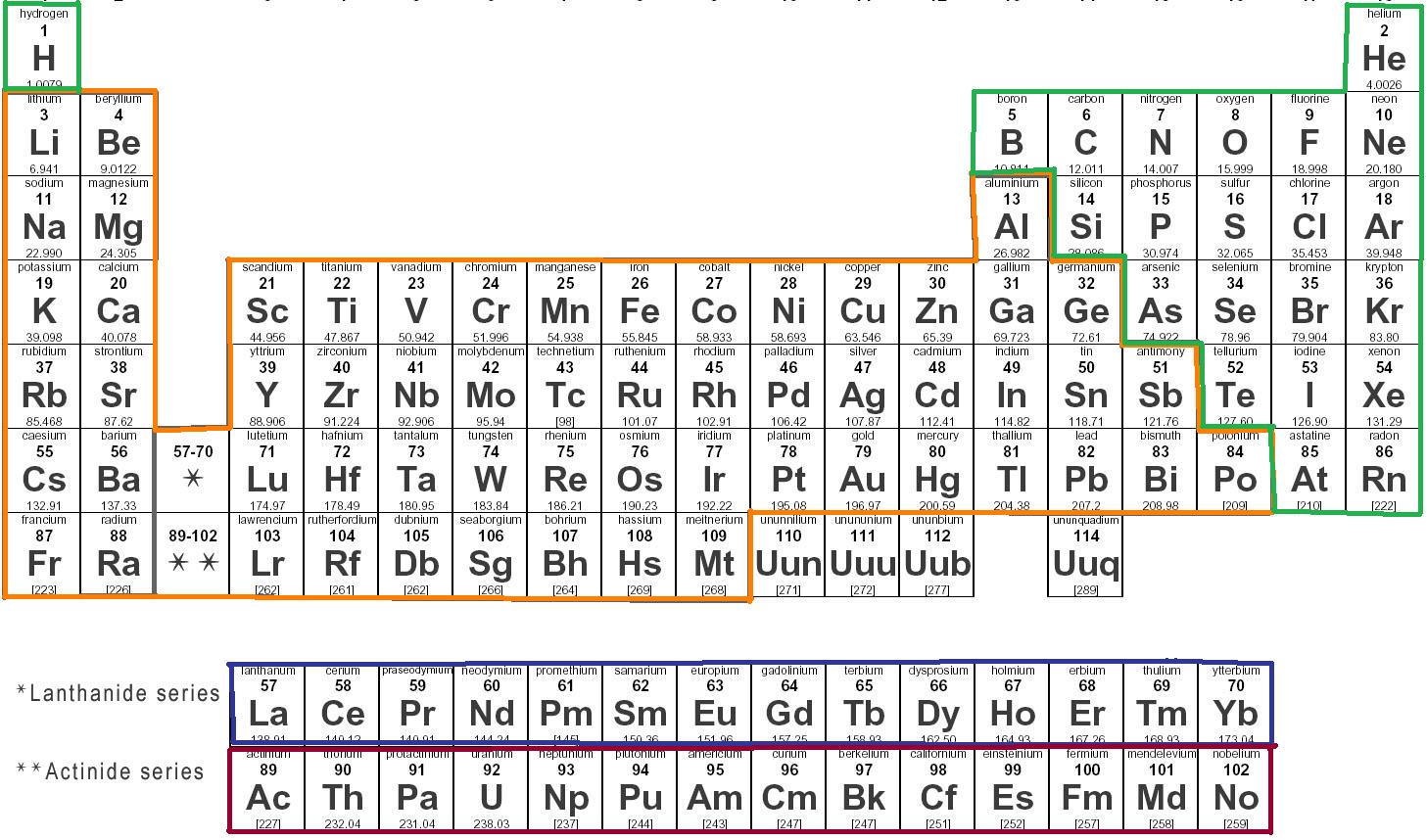
The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. Now we interpret the coefficients as referring to molar amounts, not individual molecules.) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons. We can leave out the word mol and not write the 1 coefficient (as is our habit), so the final form of the equation, still balanced, is But 6.022 × 10 23 is 1 mol, while 12.044 × 10 23 is 2 mol (and the number is written that way to make this more obvious), so we can simplify this version of the equation by writing it as It can provide you with a correct representation of any substance’ssize and mass, and valence electron shell. As a result, this chart actually displays the mass number of a specific isotope for each element. Atomic Mass Rounded Periodic Table The Routine Table is an integral part of the study of technology, and it can be useful in identifying a substance’s attributes. The lightest chemical element is Hydrogen and the heaviest is Hassium. Up to date, curated data provided by Mathematica s ElementData function from Wolfram Research, Inc. The atomic weights listed on this Table of Elements have been rounded to the nearest whole number. For chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). :max_bytes(150000):strip_icc()/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
These coefficients also have the ratio 2:1:2 (check it and see), so this equation is balanced. Notes on the properties of Silver: Specific Heat: Value given for solid phase. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
